What Roles Do Viruses Play Besides Being Pathogens?
When we think viruses, we often picture illness and disease. These microscopic entities are notorious for causing ailments like the flu or more serious conditions like HIV. However, to strictly…
Are Phages the Natural Alternatives to Antibiotics?
Antibiotic resistance is rapidly becoming one of the biggest challenges in modern medicine. As bacteria evolve and develop immunity to conventional drugs, the search for effective alternatives is more urgent…
How Viruses Can Be Friendly Helpers
When viruses are mentioned, images of flu, colds, and pandemics often come to mind. Yet, these tiny particles possess a surprisingly positive side, frequently acting as unseen allies across various…
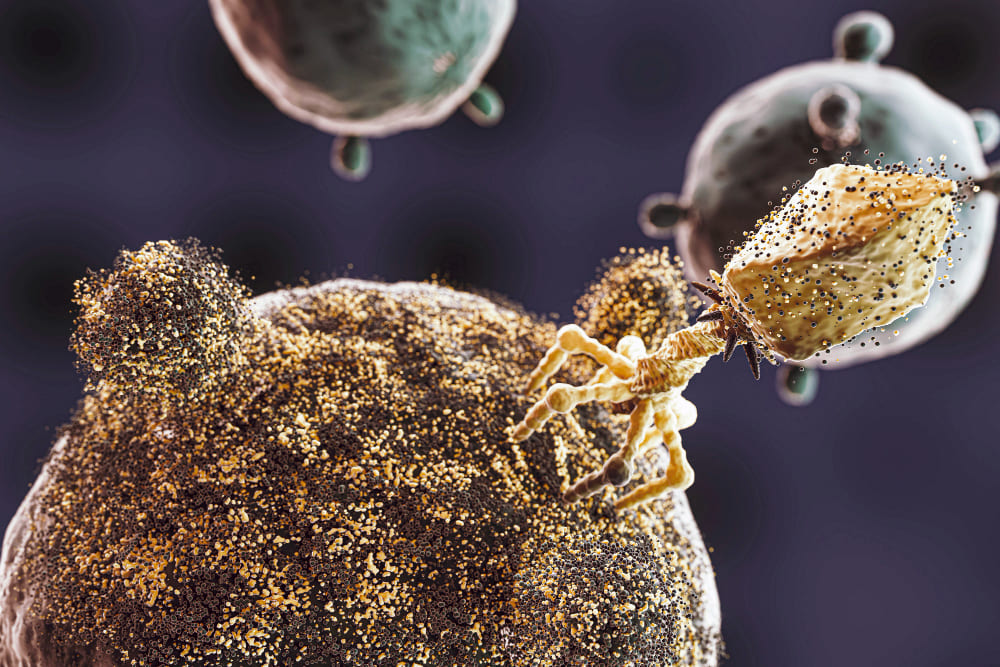
Bacteriophages: Nature’s Defense Against Bacteria
When discussing the battle against bacteria, antibiotics often steal the spotlight. However, the unsung heroes of microbial warfare are bacteriophages, or simply phages. These nifty little viruses specifically target and…